AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Carbon 14 beta decay1/8/2024  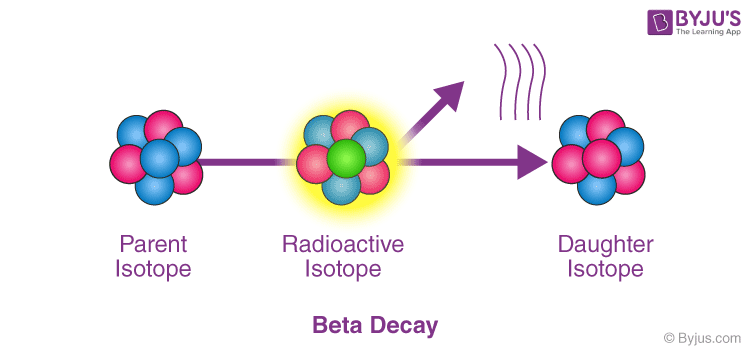
The different isotopes of carbon do not differ appreciably in their chemical properties. 
However, open-air nuclear testing between 19 contributed to this pool. The primary natural source of carbon-14 on Earth is cosmic ray action on nitrogen in the atmosphere, and it is therefore a cosmogenic nuclide. A gram of carbon containing 1 atom of carbon-14 per 10 12 atoms will emit ~0.2 beta particles per second. Carbon-14 has a maximum specific activity of 62.4 mCi/mmol (2.31 GBq/mmol), or 164.9 GBq/g. Carbon-12 and carbon-13 are both stable, while carbon-14 is unstable and has a half-life of 5700 ☓0 years. There are three naturally occurring isotopes of carbon on Earth: carbon-12 ( 12Ĭ), which makes up 99% of all carbon on Earth carbon-13 ( 13Ĭ), which makes up 1% and carbon-14 ( 14Ĭ), which occurs in trace amounts, making up about 1 or 1.5 atoms per 10 12 atoms of carbon in the atmosphere. Its existence had been suggested by Franz Kurie in 1934. 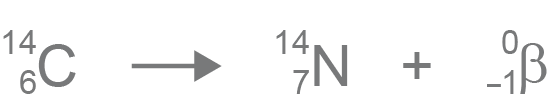
Carbon-14 was discovered on February 27, 1940, by Martin Kamen and Sam Ruben at the University of California Radiation Laboratory in Berkeley, California. Its presence in organic materials is the basis of the radiocarbon dating method pioneered by Willard Libby and colleagues (1949) to date archaeological, geological and hydrogeological samples. Carbon-14, 14Cġ part per trillion = 1 / 10 12 Ĭ or radiocarbon, is a radioactive isotope of carbon with an atomic nucleus containing 6 protons and 8 neutrons. For the dating technique, see Radiocarbon dating. For the scientific journal, see Radiocarbon (journal). After another half-life, half of what was left decays."Radiocarbon" redirects here. After one half-life, half of a sample of radioactive material has decayed. The half-life of a radioactive material can be very short (less than a second) or very long (thousands of years) or anywhere in between. Scientists use the idea of a half-life to describe this. Isotopes that do not decay are called "stable" isotopes.ĭifferent radioactive materials take different amounts of time to decay. When carbon-14 decays by emitting a beta particle, it becomes nitrogen-14. It might become a different element altogether. It may be transformed from one isotope to another. When an atom emits radiation it undergoes radioactive decay. It has 8 neutrons (instead of the usual 6) and radiates beta particles. For example, the isotope of carbon called carbon-14 is radioactive. Different isotopes have different numbers of neutrons. Most elements come in various "versions", called isotopes. It includes high energy gamma rays and X-rays. It includes alpha and beta particles as well as proton and neutron radiation. There are two main types of radiation that can be given off during radioactive decay. This process is called radioactive decay. When an atom of a radioactive substance gives off radiation, it becomes a new type of atom. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
